von Christian Fritz
We, J&K Consulting, are operating within the medicinal cannabis industry for more than one year. It all started with a coincidence – a partner asked for our GMP knowhow to process medicinal cannabis. Thus, we started following the industry, identified and analysed trends for our partners and foster continuous development. Now at the beginning of 2019 it is time to assess the activities of the past year by contrasting the divergent assumptions, techniques, and practical implementations we came across. The goal of this article is to show you the current status within the medicinal cannabis industry, as we identified many operations taking place having no real understanding of what medicinal cannabis is about and what they are actually doing. To educate we pick up eight points which we identified as core topics of matter – all connected to each other by a red line – and advise for a holistic conclusion.
1. Capital driven vs. operational driven
By legalising medicinal cannabis in Germany, we saw huge potential for the pharmaceutical industry in 2017. This also arose due to our close connection to medicinal institutions asking for a higher demand in medicinal cannabis products as of their positive influences on patients in palliative care. This was further increased by the legalisation of cannabis in Canada and within certain states of the USA in 2018. Especially, forecasts of an annual turnover of more than 20 billion Euro within an evolving industry in North America by 2020 justified a closer look. Important to mention are the different legal circumstances between the above listed countries. These information triggered interest of worldwide operating private equity groups. We were also addressed by these. However, today markets are under high pressure because of the availability of relative cheap money also for small investors, private equity was forced to invest massive amounts. This led to a capital driven situation regarding cannabis farming and production. In detail this means quick wins in generating volume of biomass were on focus while not being aware of quality. Thus, a capital driven situation is pushing the production of recreational cannabis instead of medicinal cannabis (see point 7.).
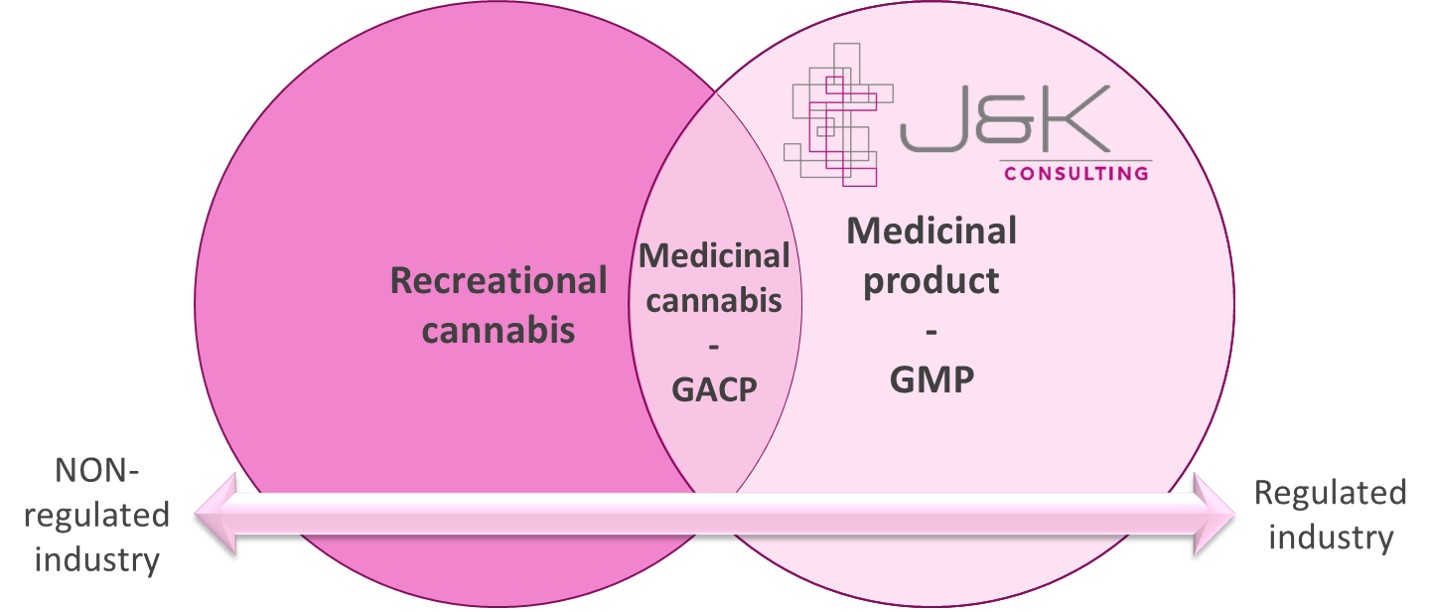
2. Medicinal cannabis vs. recreational cannabis
The economic environment and its current pointless actions endanger the potential of medicinal cannabis. Due to the existing activities it is important to highlight the differences between medicinal cannabis and recreational cannabis. The latter one is used in a broad range of products known as tobaccos or leisure/lifestyle products of the food and beverage industry. This market is strongly pushed by investments believing to produce medicinal cannabis. But this is a mistake. In detail, many and wrong media releases support creating an image of smoking dope as being representative for medicinal cannabis. The reason for this is the lack in real knowledge about “what is medicinal cannabis under the aspect of the regulated industry” and being manipulated by window dressing through marketing activities (see point 8.). To be more precisely we discussed this topic with several investors that were growing cannabis without being aware for which kind of industry they produce. All this is in contrast to medicinal cannabis used specifically for medicinal purpose since these plants need to be grown under GACP (Good Agricultural and Collection Practice) conditions. This is why the two product and thus target groups need to be strongly separated.
3. Active Pharmaceutical Ingredient (API) vs. recreational use
Separating medicinal from recreational cannabis is not just about separating product or target groups. In short, medicinal cannabis aims to be a raw material (RM) for the regulated (incl. pharmaceutical) industry. This comes along with very high standards in the area of Good Agricultural and Collection Practice (GACP) and Good Manufacturing Practice (GMP) (see point 5.). However, this RM is a source of an active pharmaceutical ingredient (API) proven by medicinal studies and triggering reactions within a human’s body. This clinical proven API effect is not the case for recreational cannabis as doses are regulated by law. Of course there are reactions due to e.g. THC or CBD but not as controlled, manageable, or proven by scientific research necessary for a pharmaceutical product. To be more precise the level, purity and quality are different and belong to very strict regulations. Additionally, be aware that the legal situation for the main ingredients of cannabis THC and CBD is varying around the globe impacting the scope and use of medicinal and recreational cannabis. Analysing our conversations with stakeholders from the existing medicinal cannabis industry made us speechless. Not even 5% were aware of the term API and what medicinal cannabis really is about.
4. ISO vs. GMP
Analysing the situation within existing production facilities or technology supplier shows that ISO is no topic in question. Also GACP and GMP seem to be answered and delivered accordingly. Nice online presentations, terms and videos pretend to be acting according to standards of the regulated industry. But going into depth (document, process and interview analyses) reveals massive lacks of knowhow and experience within the regulated industry. In practice the regulated industry is not about having general risk issues listed. It is about “how to do things” and “why you do it this way”. This leads to a risk based approach for each process. The goal is to have robust and reproduceable processes in place that ensure ongoing quality. And in case of any deviation corrective actions take place to manage issues successfully according to GMP. Thus, equipment must be qualified and processes validated according to defined specifications. Concluding, the existing medicinal cannabis industry is not able to deliver according to GACP/GMP standards.
5. Classical cultivation vs. GACP/GMP controlled cultivation
Growing cannabis is located in the horticultural sector due to its characteristics. The question is: is, according to a risk based approach (see point 4.), a classic cultivation facility like a glasshouse the right one for medicinal cannabis? Today, the cultivation taking place for cannabis equals a classic glass house production facility out of which 50% started to create a “more” controllable environment. Unfortunately, GACP and GMP criteria are rarely met. The ideal solution providing more risk control would be a fully controlled indoor cultivation facility. The challenge is to deliver equal amounts of ingredients at equal quality on a constant basis. Concluding, most facilities are under glass serving the recreational cannabis market. Thereby, these are supplied by customised technology solutions lacking in GACP and GMP knowhow, too. Summarising, due to the connection of growing cannabis as an API supplying product under a controlled environment and GACP/GMP aspects it is in question if horticultural companies performing classic cultivation are the right group of people to cultivate medicinal cannabis.
6. Light technology
A widely discussed topic for a controlled growing environment is the used light technology. Many self-named lightning experts appear on the market without any link to crowing medicinal cannabis. Two topics to be considered are light spectrum and physical appearance according to GMP guidelines. Inspecting different greenhouses indicates a lack of understanding for medicinal cannabis under the aspect of light spectrum. Mainly HPS lamps are installed to generate great biomass but do not support a controlled compound accumulation within the cannabis plant. Contrasting pest control with light technology highlights the physical appearance of a lamp. According to GACP/GMP the risk of pest control requires an easy to clean surface and purifier resistant material.
7. Push vs. pull principle
One of the best ways to understand the current cannabis business is to analyse sales. The legal changes enabled fast movers to massively push recreational cannabis products into the market as this is the easier way of production. A reason for this is the current investment environment as described under point 1 driving people to generate short term profits. What does this mean in contrast for medicinal cannabis? The answer is easy. A pull strategy driven by customer demand is required. Thus, the supply chain must be thought from a different perspective – the patients demand. In case of increasing acceptance and prove of cannabis products as a regular product in palliative care or oncology therapies this will be the driver for medicinal cannabis supply. Concluding, the medicinal cannabis market needs more activities conducted by interest groups, fist willing to promote the value of medicinal cannabis, and secondly participating as a first mover.
8. Window dressing
Linked to the pull principle is the aspect of window dressing. Recreational cannabis products are that successful, if enabled by law, because of their strong promotion. Promotion is that effective that they manage to avoid the split between recreational and medicinal cannabis. Thus, only one image of cannabis – namely recreational cannabis – is existing nowadays. Thereby medicinal cannabis seems to exist as a side product under recreational cannabis. This is a dangerous level of window dressing destroying the potential of medicinal cannabis as cannabis in general gets more and more connected to the recreational use. Additionally, this enables the cannabis market to develop apart from the regulated industry without focus on qualified facilities or validated processes according to pharmaceutical standards. To close, as people jump on this trend this leads to a vicious circle potentially destroying the huge potential of medicinal cannabis already before first potential is proven to come true.
Summarising, despite these contradictions the situation does not indicate the end of the medicinal cannabis industry. However, if not the right actions take place it will harm the European medicinal cannabis industry significantly. Therefore, we propose a holistic solution. This starts with emphasizing professional attitude, connecting reliable partners, and foster professional research leading to standardised and representative solutions for the medicinal cannabis industry.

