Our Services
- – Pharmaceutical development
- – Process-based risk management
- – Quality management
- – Project management
- – Digital Engineering


Medicinal cannabis / phytopharmaceuticals
As a consultancy firm specialising in the pharmaceutical industry, we have focused our efforts on the field of medical cannabis. We support companies in this challenging market, from the planning of state-of-the-art greenhouses and production facilities right through to the distribution of finished cannabis-based medicines.
Our aim is to uphold the highest pharmaceutical quality standards and to work with you to develop high-quality medicinal cannabis products.
As a consultancy firm specialising in the pharmaceutical industry, we have focused our efforts on the field of medical cannabis. We support companies in this challenging market, from the planning of state-of-the-art greenhouses and production facilities right through to the distribution of finished cannabis-based medicines.
Our aim is to uphold the highest pharmaceutical quality standards and to work with you to develop high-quality medicinal cannabis products.
Achievements and references
Our wide-ranging projects across various companies are based on high-quality consultancy. We take the time to work with each individual client to analyse challenges in detail and then tackle them successfully.
Please feel free to take a look at our references and project examples.


Publications
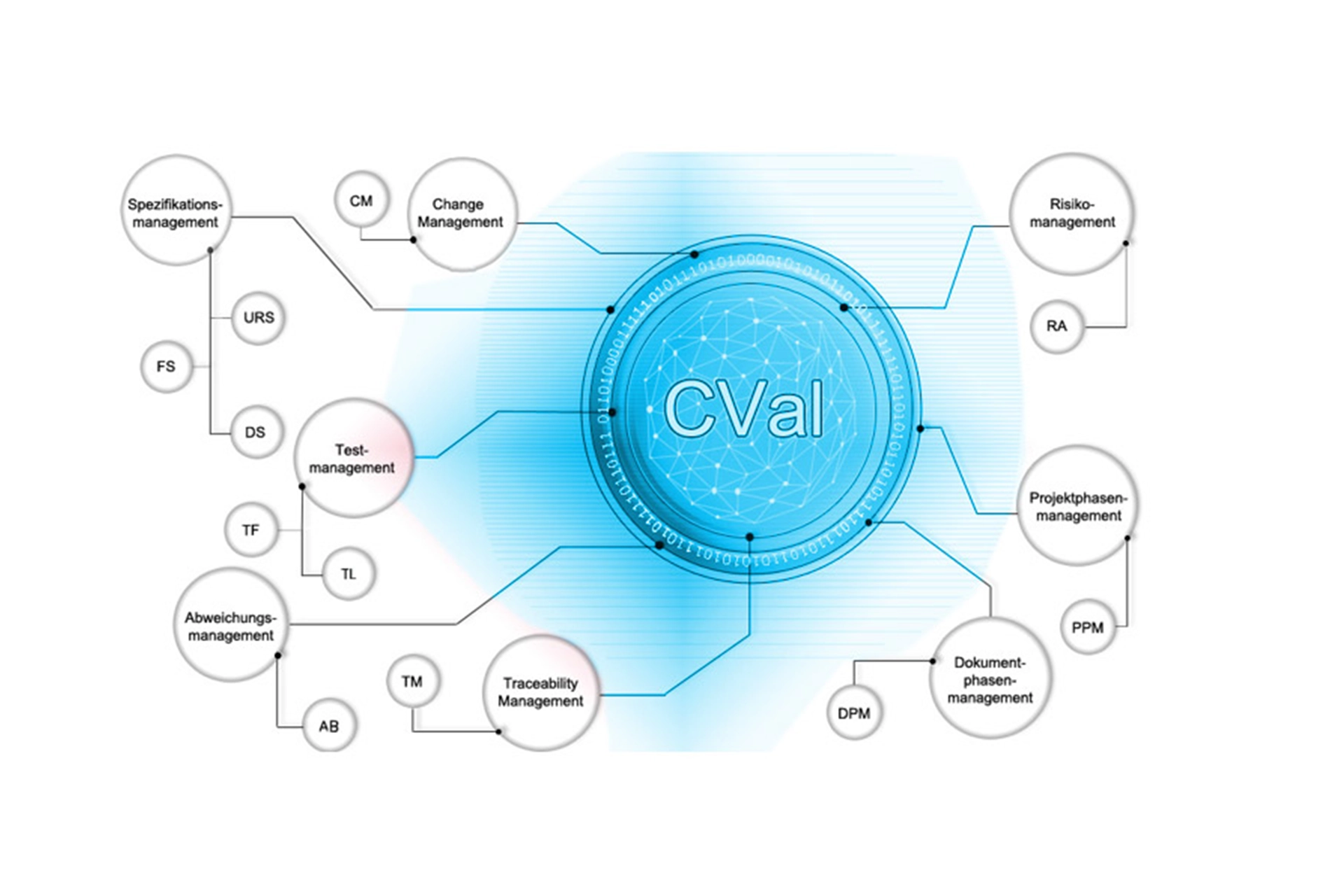
CVal